
Proton Exchange Membrane Water Electrolyser (PEMWE)
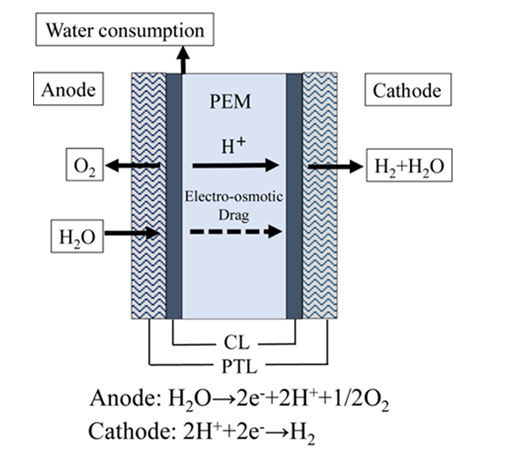
Proton Exchange Membrane Water Electrolysers (PEMWEs) split water into hydrogen and oxygen in the presence of an electric current. They utilise a solid polymer membrane (electrolyte), which facilitates the transfer of protons from the anode to the cathode. At the anode, water oxidises to produce oxygen, protons, and electrons. The protons migrate through the polymer membrane to the cathode, where they combine with the electrons to produce hydrogen. If the electricity is renewable, green hydrogen with no emissions will be produced. PEMWEs are hailed as the future of green hydrogen, due to their high efficiencies, high hydrogen purity, compact design, and relatively low operating temperatures. Yet, their biggest advantage lies in their ability to quickly adjust to power supply fluctuations, making for an ideal integration with intermittent renewable electricity sources, such as wind and solar energy. Furthermore, PEMWE is the only electrolyser that produces dry hydrogen. Hence, an extra dryer module is not needed to separate the water, allowing for direct storage and utilisation.
Spurred by governments scrambling to establish themselves s leaders in a green hydrogen economy, successful pilot demonstrations, and initial commercialisation, the global PEMWE market size reached £6.47B in 2023, and is poised for exponential market growth, reaching £47.5B by 2032 (Global Market Insights). This growth projection is backed by strong policy and investment decisions, namely the UK Net Zero Hydrogen Fund, US Department of Energy H2Hubs, and the European Green Deal and Hydrogen Strategy for a Climate-Neutral Europe. PEMWE electrolysers are being hailed as the future of low-carbon hydrogen production, yet key issues are preventing widespread adoption. The materials used for electrodes are normally precious metals, significantly increasing the cost. The lifespan of PEMWE stacks also remains an issue, with significant degradation observed compared to Alkaline Electrolysers. Questions about the scalability and cost-effectiveness of PEMWEs need to be addressed before widespread adoption.
Challenges Revealed Through Literature Review
BiPolar Plates
- Reduce the use of high-cost substrates
- Suppression of quality variation
- Surface coating technology to reduce precious metal usage
- Anode dissolution-resistant materials
- Cathode hydrogenation resistance
- Comprehensive evaluation criteria for degradation (constant/changing load)
- Porous Transport Layer
- Reduce the use of high-cost substrates (RtR continuous production process)
- Develop technology to suppress quality variation
- Structural optimization using multi-short design simulation etc.
- Achieve a balance between usage and durability of high-cost base materials (e.g. Ti)
- MEA (CCM) short circuit prevention
- Elucidate the PTL degradation mechanism (constant load → changing load, etc.)
- Elucidate the bubble ejection mechanism (pore size distribution and changes, etc.)
- Anode/Cathode Catalyst Layer
- Reduce the use of precious metals and improve material durability
- Improve durability during load changes and start/stop mechanisms
- Reduce use of (find alternatives) precious metals (Ti/Ir for anode, Pt for cathode), including alloys
- Elucidate the catalyst degradation mechanism (evaluation standardisation, varying load, etc.)
Electrolyte
- Suppression of quality variation
- Improve mechanical properties (withstand pressure difference)
- Achieve high current density, safety, reliability, high availability, low cost substrates
- Improve durability during load fluctuation operation and start-stop operation
- Inhibit chemical deterioration (e.g. free radical resistance of the film)
- Elucidate electrolyte membrane degradation (constant load, changing load)
Membrane Electrode Assembly
- Structure optimisation
- Elucidate the phenomena of moisture, gas and proton transport in the catalyst layer
- Drying process and mass production
- Improve hydrogen conversion efficiency and hydrogen production per cell unit area
- High speed, high precision additive manufacturing
- Reduce contact resistance between components, part count, assembly time
- Ensure the uniformity of components within the plane and battery pack
- Reduce temperature changes and improve cell sealing within the battery
- Develop automatic manufacturing/assembly technology
- Modular standardisation
- Ensure uniform electrochemical reaction and material diffusion within the plane and within the stack
- Simple calculation method to optimize fastening load distribution and resistance
Academic Capability Mapping

Word cloud
The word-cloud of the primary and secondary keywords is presented for the Proton Exchange Membrane Water Electrolyser (PEMWE) technology. These keywords were used as the input to Scopus for the purpose of the Academic Capability Mapping. The analysis underscores key research areas like electrocatalysis and membranes.
Documents by Country
The number of papers published worldwide pertaining to Proton Exchange Membrane Water Electrolysers (PEMWE) since the year 2000, divided into three decades. Only the top 10 countries are displayed. It is interesting to note that the UK is number 8 in PEMWE research, despite common misconceptions that the UK has strong academic capability in the field of electrochemistry.
Documents by Author (2000 – 2025)
Prominent UK academics and their affiliation is showcased. The y-axis represents the H-index of the authors, while the x-axis illustrates the number of papers published. It can be clearly seen that while the UK authors may have high H-index’s, the number of papers in the field of PEMWE are less than 7.
Documents by Affiliation
The number of papers published by affiliation in the UK since the year 2000 are showcased. The University of Glasgow leads the way with the most publications, closely followed by Newcastle University. The figure specifically highlights the top 10 UK institutions in the field of Proton Exchange Membrane Water Electrolysers, providing a definite ranking list of universities with excellent expertise in the PEMWE technology.
Proton Exchange Membrane Water Electrolyser (PEMWE) – Delphi Survey Analysis
Participant Identifiers
Industry Collaboration
Confidence Level
Country Affiliation
Key Performance Indicators
Key technical target predictions were provided by the participants, expected to be achieved by 2030.
Cell Level KPIs
Stack and System KPIs
Challenges
The participants were provided with several options and were asked to rank these options from 0 (least critical) to 6 (most critical). They were also provided with a text option to suggest additional challenges.
Bi-Polar Plates
Electrolyte
Stack and System
Membrane Electrode Assembly
Porous Transport Layer
Anode Catalyst Layer
Proton Exchange Membrane Water Electrolyser System Integration
- Balancing intermittent renewable generation
- Grid stability through adjustable hydrogen production rates
- Peak regulation and voltage regulation
- Energy conversion and storage
- Chemical plants for H2 utilisation
- Ammonia synthesis
- Metal refining
- Steel production
- Solar, wind, or biomass facilities supplying electricity
- Infrastructure to ensure a consistent water supply
- Bulk power grid
- Power quality
- Hydrogen storage and transport facilities
- Synthetic ammonia installation
- Fuel Cells
- Gas dehumidification and compression